|
R&D Systems
mouse timp 2 concentration Mouse Timp 2 Concentration, supplied by R&D Systems, used in various techniques. Bioz Stars score: 92/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/mouse timp 2 concentration/product/R&D Systems Average 92 stars, based on 1 article reviews
mouse timp 2 concentration - by Bioz Stars,
2026-03
92/100 stars
|
Buy from Supplier |
|
Bio-Techne corporation
recombinant mouse timp2 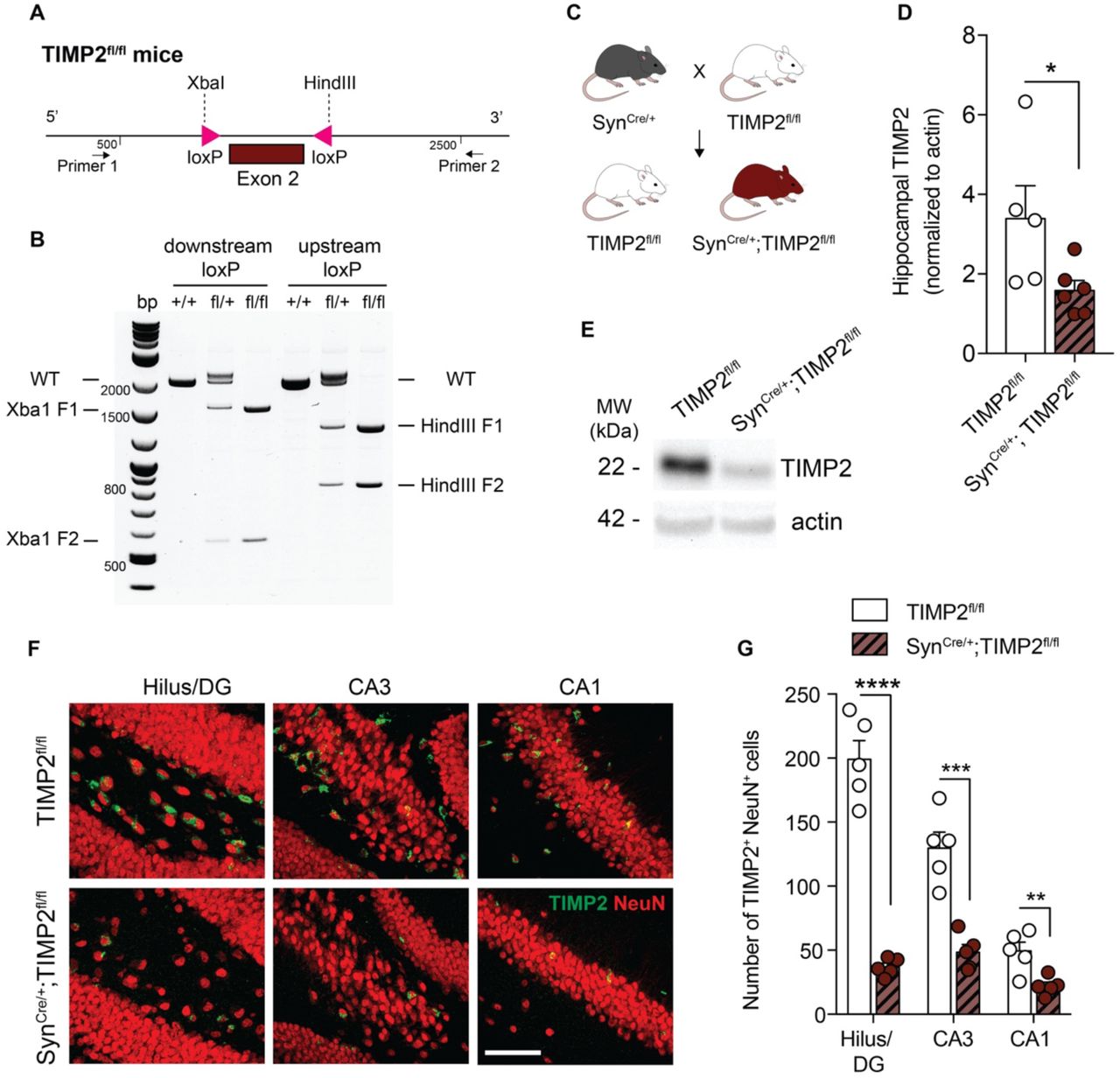 Recombinant Mouse Timp2, supplied by Bio-Techne corporation, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/recombinant mouse timp2/product/Bio-Techne corporation Average 90 stars, based on 1 article reviews
recombinant mouse timp2 - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
R&D Systems
mouse timp2  Mouse Timp2, supplied by R&D Systems, used in various techniques. Bioz Stars score: 92/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/mouse timp2/product/R&D Systems Average 92 stars, based on 1 article reviews
mouse timp2 - by Bioz Stars,
2026-03
92/100 stars
|
Buy from Supplier |
|
R&D Systems
human timp2 levels  Human Timp2 Levels, supplied by R&D Systems, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/human timp2 levels/product/R&D Systems Average 93 stars, based on 1 article reviews
human timp2 levels - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
R&D Systems
timp 2 concentrations  Timp 2 Concentrations, supplied by R&D Systems, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/timp 2 concentrations/product/R&D Systems Average 93 stars, based on 1 article reviews
timp 2 concentrations - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
R&D Systems
mmp2  Mmp2, supplied by R&D Systems, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/mmp2/product/R&D Systems Average 93 stars, based on 1 article reviews
mmp2 - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
Proteintech
timp 2  Timp 2, supplied by Proteintech, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/timp 2/product/Proteintech Average 93 stars, based on 1 article reviews
timp 2 - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
R&D Systems
timp2  Timp2, supplied by R&D Systems, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/timp2/product/R&D Systems Average 93 stars, based on 1 article reviews
timp2 - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
R&D Systems
elisas  Elisas, supplied by R&D Systems, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/elisas/product/R&D Systems Average 93 stars, based on 1 article reviews
elisas - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
R&D Systems
timp 2  Timp 2, supplied by R&D Systems, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/timp 2/product/R&D Systems Average 93 stars, based on 1 article reviews
timp 2 - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
R&D Systems
mouse timp-2 duoset enzyme-linked immunosorbent assay (elisa) kit  Mouse Timp 2 Duoset Enzyme Linked Immunosorbent Assay (Elisa) Kit, supplied by R&D Systems, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/mouse timp-2 duoset enzyme-linked immunosorbent assay (elisa) kit/product/R&D Systems Average 90 stars, based on 1 article reviews
mouse timp-2 duoset enzyme-linked immunosorbent assay (elisa) kit - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Millipore
monoclonal antibody anti-timp-2 mouse (mab t2-101)  Monoclonal Antibody Anti Timp 2 Mouse (Mab T2 101), supplied by Millipore, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/monoclonal antibody anti-timp-2 mouse (mab t2-101)/product/Millipore Average 90 stars, based on 1 article reviews
monoclonal antibody anti-timp-2 mouse (mab t2-101) - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
Image Search Results

Journal: bioRxiv
Article Title: Neuronal TIMP2 regulates hippocampus-dependent plasticity and extracellular matrix complexity
doi: 10.1101/2022.12.28.522138
Figure Lengend Snippet: (A) Schematic illustration of the targeting strategy of to insert loxP sites flanking exon 2 to generate a model for conditional deletion of TIMP2 . (B) Representative genotyping results revealing the PCR products for the mutant LoxP and wild-type alleles. Wild-type (+/+), heterozygous (fl/+), and homozygous (fl/fl) mice were identified according to this strategy. (C) Schematic diagram of cross-breeding strategy to establish neuron-specific TIMP2 deletion: male TIMP2 fl/fl mice were mated with Syn Cre/+ females to obtain Syn Cre/+ ; TIMP2 fl/fl and their respective TIMP2 fl/fl littermate controls. (D-E) Representative TIMP2 immunoblot and corresponding quantification of TIMP2 protein levels from hippocampal lysate of TIMP2 fl/fl and Syn Cre/+ ; TIMP2 fl/fl (2-3 months of age, N = 5-6 mice per group). (F) High-magnification view of hilus/DG, CA3, and CA1 sub-regions of TIMP2+ cells co-expressing NeuN in TIMP2 fl/fl and Syn Cre/+ ; TIMP2 fl/fl mice (2-3 months of age, N = 5 mice per group, scale bar, 20 μm) with corresponding (G) quantification of the total number of TIMP2+ cells with NeuN+ nuclei across hippocampal subregions. Data are represented as mean ± SEM. Student’s t-test for two-group comparisons. * P <0.05, ** P <0.01, *** P <0.001, **** P <0.0001. Data points represent individual mice.
Article Snippet: Estimation of eISF TIMP2 protein levels was determined by extrapolation from a standard curve of
Techniques: Mutagenesis, Western Blot, Expressing

Journal: bioRxiv
Article Title: Neuronal TIMP2 regulates hippocampus-dependent plasticity and extracellular matrix complexity
doi: 10.1101/2022.12.28.522138
Figure Lengend Snippet: (A) Low-magnification view (upper image) of mouse hippocampus and high-magnification view (lower images) of hilus/DG, CA3, and CA1 subregions showing TIMP2 + and TIMP2 + NeuN + cells. Scale bars, 200 μm and 20 μm (inset). (B) Quantification of the total number of TIMP2 + cells and TIMP2 + NeuN + cells across hippocampal subregions in WT mice (2 months of age; N = 8, males and females). (C) Schematic representation of high molecular-weight cut-off (1-MDa) in vivo microdialysis to assay TIMP2 levels in mouse hippocampal ISF. (D) TIMP2 immunoblotting of hippocampal ISF dialyzed from 2-month-old WT and TIMP2 KO mice, with corresponding Ponceau S stain. (E) Schematic representation of bulk RNA-seq workflow performed in isolated WT and TIMP2 KO hippocampi (N=13-17/group, sex-matched) for transcriptomic analysis. (F) Volcano plot showing the fold-change of genes (log 2 scale) differentially expressed in hippocampus of TIMP2 KO vs. WT mice. Downregulated DEGs at P < 0.05 are highlighted in red (upregulated in black). (G) Top 10 significant pathways for downregulated DEGs from Gene Set Enrichment Analysis. Data are represented as mean ± SEM. DG, dentate gyrus; eISF, exchangeable interstitial fluid; aCSF, artificial cerebrospinal fluid.
Article Snippet: Estimation of eISF TIMP2 protein levels was determined by extrapolation from a standard curve of
Techniques: Molecular Weight, In Vivo, Western Blot, Staining, RNA Sequencing Assay, Isolation

Journal: bioRxiv
Article Title: Neuronal TIMP2 regulates hippocampus-dependent plasticity and extracellular matrix complexity
doi: 10.1101/2022.12.28.522138
Figure Lengend Snippet: (A) Quantification of the total number of TIMP2 + and TIMP2 + NeuN + cells at the hilus/DG, (B) CA3 and (C) CA1 subregions of the hippocampus from WT males and females at 2 months of age (N = 4 mice per sex). (D) Cresyl violet-stained section depicting the microdialysis probe tract through brain surface extending into hippocampus, with microdialysis probe position seen at Bregma - 3.08mm based on Paxinos and Watson atlas. Scale bar, 500 μm. (E) Top 10 significant pathways for upregulated DEGs in hippocampi of TIMP2 KO mice relative to WT mice (N = 13-17 mice per group). Data are represented as mean ± SEM.
Article Snippet: Estimation of eISF TIMP2 protein levels was determined by extrapolation from a standard curve of
Techniques: Staining

Journal: bioRxiv
Article Title: Neuronal TIMP2 regulates hippocampus-dependent plasticity and extracellular matrix complexity
doi: 10.1101/2022.12.28.522138
Figure Lengend Snippet: (A) Schematic timeline of BrdU intraperitoneal injection protocol to label proliferating cells in the DG in isolated brain sections. (B) Schematic timeline of BrdU intraperitoneal injection protocol used for cell fate “survival” labeling in DG of isolated brain sections. (C) Representative confocal microscopy images of BrdU + cells in the DG of WT and TIMP2 KO mice (2-3 months of age, N = 11-12 mice per group; arrowheads indicate BrdU + cells; scale bar, 50 μm) with corresponding (D) quantification of number of BrdU + cells in the dentate gyrus per unit area. (E) Representative confocal microscopy images of proliferating Ki67 + cells in the DG of WT and TIMP2 KO mice (2-3 months of age, N = 11-12 mice per group; arrowheads indicate Ki67 + cells; scale bar, 50 μm) with corresponding (F) quantification of the number of Ki67 + proliferating cells per DG in WT and TIMP2 KO mice. (G) Representative confocal microscopy images of Sox2 + in the DG of WT and TIMP2 KO mice (2-3 months of age, N = 11-12 mice per group; arrowheads indicate Sox2 + cells; scale bar, 50 μm) with corresponding (H) quantification of Sox2 + neural progenitor cells in the DG of WT and TIMP2 KO mice. (I) Representative confocal microscopy images of DCX + cells in the DG of WT and TIMP2 KO mice (2-3 months of age, N = 11-12 mice per group; scale bar, 50 μm) with corresponding (J) quantification of DCX + immature neuroblasts in DG of WT and TIMP2 KO mice. (K) Representative confocal microscopy images of BrdU + NeuN + cells in DG of WT and TIMP2 KO mice (2-3 months of age. N = 8-11 mice per group; scale bar, 50 μm and 100 μm (inset)) with corresponding (L) quantification of newborn neurons (BrdU + NeuN + cells) in the DG of WT and TIMP2 KO mice. Data are represented as mean ± SEM. Student’s t -test for two-group comparisons. * P <0.05; ** P <0.01; *** P <0.001. Data points represent individual mice. IHC, immunohistochemistry; DG, dentate gyrus; DCX, doublecortin.
Article Snippet: Estimation of eISF TIMP2 protein levels was determined by extrapolation from a standard curve of
Techniques: Injection, Isolation, Labeling, Confocal Microscopy, Immunohistochemistry

Journal: bioRxiv
Article Title: Neuronal TIMP2 regulates hippocampus-dependent plasticity and extracellular matrix complexity
doi: 10.1101/2022.12.28.522138
Figure Lengend Snippet: (A) Schematic representation of the workflow for dendritic spine quantification using Lucifer Yellow-filled DG granule cells. (B) Overall dendritic spine density in DG granule cells iontophoretically labeled with Lucifer Yellow using sections isolated from six-month-old WT and TIMP2 KO mice (N=6 neurons per mouse from N=4-5 mice per group). (C) Quantification of the percentage of spines categorized according to “thin” spine heads or (D) “mushroom” spine heads. (E) Representative deconvolved confocal image of a dendritic segment from WT and TIMP2 KO Lucifer Yellow-labeled brain sections, and the downstream 3D reconstruction, with dendritic segment shown in pink, thin spines in green, stubby in blue, mushroom in red, and filopodia in yellow. Scale bar, 2 μm. Data are represented as mean ± SEM. Nested t-test for comparisons with neuron and mouse as levels. * P <0.05; ** P <0.01. Data points represent neurons (left) and mice (right) for each group. LY, Lucifer Yellow.
Article Snippet: Estimation of eISF TIMP2 protein levels was determined by extrapolation from a standard curve of
Techniques: Labeling, Isolation

Journal: bioRxiv
Article Title: Neuronal TIMP2 regulates hippocampus-dependent plasticity and extracellular matrix complexity
doi: 10.1101/2022.12.28.522138
Figure Lengend Snippet: (A) Schematic of the novel location recognition assay. (B) Discrimination index for novel location recognition on day 2 for WT and TIMP2 KO mice (2-3 months of age, N = 11-12 mice per group). (C) Schematic of the contextual and cued fear-conditioning assay. (D, E) Freezing levels by interval and overall measured in the conditioned-fear context A in WT and TIMP2 KO mice (2-3 months of age, N = 11-12 mice per group). (F) Schematic diagram of modified Barnes maze with color coding for overall strategy classification. (G) Proportion of WT and TIMP2 KO mice using non-hippocampus-dependent (gray) and hippocampus-dependent (yellow) strategies during the testing trials (2-3 months of age, n = 9 mice per group). (H) Strategy utilization by WT and TIMP2 KO mice on day 3 of the Barnes Maze and corresponding (I) cognitive scores, ranked by strategy complexity for WT and TIMP2 KO mice on day 3 in the Barnes Maze. Data are represented as mean ± SEM. Student’s t-test for two-group comparisons in (B, D, E) , chi-square test in (H) , and nested t-test (I) for trial and mouse levels. * P <0.05, ** P <0.01. Data points represent individual mice.
Article Snippet: Estimation of eISF TIMP2 protein levels was determined by extrapolation from a standard curve of
Techniques: Modification

Journal: bioRxiv
Article Title: Neuronal TIMP2 regulates hippocampus-dependent plasticity and extracellular matrix complexity
doi: 10.1101/2022.12.28.522138
Figure Lengend Snippet: (A) Percentage of freezing detected in the cued task of the fear-conditioning assay in WT and TIMP2 KO mice (2-3 months of age, N = 12 mice per group). (B) Latency of TIMP2 KO and WT mice to fall in the rotarod in the fixed and (C) acceleration protocol at 4, 20 and 40 rpm (N = 12 mice per group). (D) Total distance traveled by TIMP2 KO and WT mice in the open field, as well as (E) velocity, and (F) percentage of time spent in the center of the arena (N = 11-12 mice per group). (G) Latency of TIMP2 KO and WT mice to fall in the wire test (N = 12 mice per group). (H) Time for TIMP2 KO and WT mice to descend the pole in the pole test (N = 11-12 mice per group). (I) Grip strength of the fore-, hind- and four limbs in TIMP2 KO and WT mice (N = 12 mice per group). (J) Hindlimb extension by clasping score in TIMP2 KO and WT mice (N = 10-11 mice per group). Data are represented as mean ± SEM. Student’s t -test for two-group comparisons. n.s., not significant.
Article Snippet: Estimation of eISF TIMP2 protein levels was determined by extrapolation from a standard curve of
Techniques:

Journal: bioRxiv
Article Title: Neuronal TIMP2 regulates hippocampus-dependent plasticity and extracellular matrix complexity
doi: 10.1101/2022.12.28.522138
Figure Lengend Snippet: (A) MMP2 mRNA levels by qPCR from isolated hippocampus of WT and TIMP2 KO mice (2-3 months of age, N = 6 mice per group). (B) Immunoblotting of MMP2 from hippocampal lysates of WT and TIMP2 KO mice (2-3 months of age, N = 9 mice per group) with corresponding (C) quantification of MMP2 protein levels. (D) Representative confocal microscopy images showing aggrecan puncta in the molecular layer of DG from WT and TIMP2 KO mice (2-3 months of age, N = 8-13 mice per group; scale bar, 5 μm) with corresponding (E) quantification of puncta. (F) Representative confocal microscopy images and corresponding (G) quantification of aggrecan and homer1 co-localization in the molecular layer of DG from WT and TIMP2 KO mice (2-3 months of age, N = 8-13 mice per group; scale bar, 5 μm). Circles indicate colocalized puncta. (H) Representative images showing the migration of DCX + cells in the SGZ and GCL of WT and TIMP2 KO mice (2-3 months of age, N = 11-12 mice per group, scale bar, 20 μm with corresponding (I) quantification of DCX + cells distributed in the subgranular zone (SGZ) and granule cell layer (GCL) of DG. (J) Representative surface scanning electron microscopy (SEM) from DG of WT and TIMP2 KO mice (2-3 months of age, N = 3 mice per group, scale bar, 2 μm, with corresponding (K) quantification of ECM microarchitecture in terms of fiber diameter measurements. Data are represented as mean ± SEM. Student’s t-test for two-group comparisons. * P <0.05, ** P <0.01, *** P <0.001. Data points represent individual mice.
Article Snippet: Estimation of eISF TIMP2 protein levels was determined by extrapolation from a standard curve of
Techniques: Isolation, Western Blot, Confocal Microscopy, Migration, Electron Microscopy

Journal: bioRxiv
Article Title: Neuronal TIMP2 regulates hippocampus-dependent plasticity and extracellular matrix complexity
doi: 10.1101/2022.12.28.522138
Figure Lengend Snippet: (A) Discrimination index for novel location recognition on day 2 and (B,C) contextual fear-conditioning freezing levels in TIMP2 fl/fl and Syn Cre/+ ; TIMP2 fl/fl mice (2-3 months of age, N = 13-15 mice per group). (D) Proportion of mice of each genotype using non-hippocampus-dependent (gray) and hippocampus-dependent (yellow) search strategies during the testing trials of TIMP2 fl/fl and Syn Cre/+ ; TIMP2 fl/fl mice in Barnes maze (2-3 months of age, N = 12-14 mice per group) with (E) quantification of the percentage of mice using these strategies by on day 3. (F) Cognitive complexity scores for TIMP2 fl/fl and Syn Cre/+ ; TIMP2 fl/fl mice based on strategies used on day 3 of Barnes maze. (G) Representative confocal microscopy images of BrdU + and NeuN + cells in DG of TIMP2 fl/fl and Syn Cre/+ ; TIMP2 fl/fl mice (2-3 months of age, N = 7–8 mice per group, scale bar, 100 μm) with corresponding (H) quantification of BrdU + NeuN + newborn neurons in the DG. (I) Representative confocal microscopy images of aggrecan and homer1 puncta in molecular layer of DG of TIMP2 fl/fl and Syn Cre/+ ; TIMP2 fl/fl mice (N = 7-8 mice per group, scale bar, 5 μm) with corresponding (J) quantification of aggrecan puncta and (K) co-localized puncta of aggrecan and homer1, as indicated by overlaid circles. Data are represented as mean ± SEM. Student’s t -test for two-group comparisons in (A-C) , (H) , and (J, K) , chi-square test in (E) , and nested t-test (F) with trial and mice as levels. * P <0.05, ** P <0.01, *** P <0.001, **** P <0.0001. Data points represent individual mice.
Article Snippet: Estimation of eISF TIMP2 protein levels was determined by extrapolation from a standard curve of
Techniques: Confocal Microscopy

Journal: bioRxiv
Article Title: Neuronal TIMP2 regulates hippocampus-dependent plasticity and extracellular matrix complexity
doi: 10.1101/2022.12.28.522138
Figure Lengend Snippet: (A) Percentage of freezing detected in the cued task of the fear-conditioning assay in TIMP2 fl/fl and Syn Cre/+ ; TIMP2 fl/fl mice (2-3 months of age, N = 13-15 mice per group). (B) Latency of TIMP2 fl/fl and Syn Cre/+ ; TIMP2 fl/fl mice to fall in the rotarod in the fixed and (C) acceleration protocol at 4, 10 and 20 rpm (N = 13-15 mice per group). (D) Total distance traveled by TIMP2 fl/fl and Syn Cre/+ ; TIMP2 fl/fl mice in the open field, as well as (E) velocity, and (F) percentage of time spent in the center of the arena (N = 13-15 mice per group). (G) Latency of TIMP2 fl/fl and Syn Cre/+ ; TIMP2 fl/fl mice to fall in the wire test (N = 12-14 mice per group). (H) Time for TIMP2 fl/fl and Syn Cre/+ ; TIMP2 fl/fl mice to descend the pole in the pole test (N = 12-14 mice per group). (I) Grip strength of the fore-, hind- and four limbs in TIMP2 fl/fl and Syn Cre/+ ; TIMP2 fl/fl mice (N = 13-15 mice per group). (J) Hindlimb extension by clasping score in TIMP2 fl/fl and Syn Cre/+ ; TIMP2 fl/fl mice (N = 12-14 mice per group). Data are represented as mean ± SEM. Student’s t -test for two-group comparisons. n.s., not significant.
Article Snippet: Estimation of eISF TIMP2 protein levels was determined by extrapolation from a standard curve of
Techniques:

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: Key resources
Article Snippet: Response 3: To measure
Techniques: Recombinant, Diagnostic Assay, Plasmid Preparation, Staining, Extraction, Sterility, Saline, Construct, Electron Microscopy, Expressing, Enzyme-linked Immunosorbent Assay, SYBR Green Assay, Multiplex Assay, Transfection, Sequencing, Software, Control

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: Characterization of TIMP2 protein constructs
Article Snippet: Response 3: To measure
Techniques: Concentration Assay

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: Description of mouse cohorts
Article Snippet: Response 3: To measure
Techniques:

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: Hippocampal gene expression following treatment with TIMP2 and TIMP2-hIgG4
Article Snippet: Response 3: To measure
Techniques: Expressing

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: Few significant changes in hippocampal gene expression following treatment with TIMP2 or TIMP2-hIgG4
Article Snippet: Response 3: To measure
Techniques: Expressing

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: The TIMP2 constructs had distinct MMP inhibitory profiles
Article Snippet: Response 3: To measure
Techniques: Construct, Inhibition

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: The alanine insertion into TIMP2 prevented MMP inhibitory activity at biologically relevant concentrations
Article Snippet: Response 3: To measure
Techniques: Activity Assay

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: MMP protein constructs. Schematic depicting the nine MMP protein constructs assessed for binding to TIMP2 constructs using bio-layer interferometry (BLI) and surface plasmon resonance (SPR). CD, catalytic domain.
Article Snippet: Response 3: To measure
Techniques: Construct, Binding Assay, SPR Assay

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: Characterization of TIMP2-MMP binding of the TIMP2 constructs
Article Snippet: Response 3: To measure
Techniques: Binding Assay

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: Key resources
Article Snippet:
Techniques: Recombinant, Diagnostic Assay, Plasmid Preparation, Staining, Extraction, Sterility, Saline, Construct, Electron Microscopy, Expressing, Enzyme-linked Immunosorbent Assay, SYBR Green Assay, Multiplex Assay, Transfection, Sequencing, Software, Control

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: Characterization of TIMP2 protein constructs
Article Snippet:
Techniques: Concentration Assay

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: Description of mouse cohorts
Article Snippet:
Techniques:

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: Hippocampal gene expression following treatment with TIMP2 and TIMP2-hIgG4
Article Snippet:
Techniques: Expressing

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: Few significant changes in hippocampal gene expression following treatment with TIMP2 or TIMP2-hIgG4
Article Snippet:
Techniques: Expressing

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: The TIMP2 constructs had distinct MMP inhibitory profiles
Article Snippet:
Techniques: Construct, Inhibition

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: The alanine insertion into TIMP2 prevented MMP inhibitory activity at biologically relevant concentrations
Article Snippet:
Techniques: Activity Assay

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: MMP protein constructs. Schematic depicting the nine MMP protein constructs assessed for binding to TIMP2 constructs using bio-layer interferometry (BLI) and surface plasmon resonance (SPR). CD, catalytic domain.
Article Snippet:
Techniques: Construct, Binding Assay, SPR Assay

Journal: eNeuro
Article Title: Noncanonical Activity of Tissue Inhibitor of Metalloproteinases 2 (TIMP2) Improves Cognition and Synapse Density in Aging
doi: 10.1523/ENEURO.0031-23.2023
Figure Lengend Snippet: Characterization of TIMP2-MMP binding of the TIMP2 constructs
Article Snippet:
Techniques: Binding Assay

Journal: Haematologica
Article Title: Endotoxemia shifts neutrophils with TIMP-free gelatinase B/MMP-9 from bone marrow to the periphery and induces systematic upregulation of TIMP-1
doi: 10.3324/haematol.2017.168799
Figure Lengend Snippet: Systematic induction of TIMP-1 and reduced bone marrow MMP gelatinolytic activity. (A) TIMP-1 and TIMP-2 protein content in bone marrow, spleen, lungs and liver of mice injected i.p. with LPS or control mice (PBS injection) as determined by ELISA. Protein levels detected by ELISA were corrected for the total protein concentration and are presented as nanograms of TIMP in one milligram of total protein. (B) Degradation of fluorogenic gelatin by gelatinases present in bone marrow samples from mice treated with LPS and control mice, in the presence or absence of an inhibitor of MMP-2 and MMP-9 (SB-3CT, 10 μM) or elastase inhibitor (ElaV, 10 μM). The velocity of the gelatin degradation reaction was expressed as fluorescence units (FU) per minute and is indicative of the net proteolytic activity present in the samples. Inhibition percentages are shown. * P <0.05, ** P <0.01, as determined by the Mann-Whitney test.
Article Snippet: TIMP-1 and
Techniques: Activity Assay, Injection, Enzyme-linked Immunosorbent Assay, Protein Concentration, Fluorescence, Inhibition, MANN-WHITNEY

Journal: Haematologica
Article Title: Endotoxemia shifts neutrophils with TIMP-free gelatinase B/MMP-9 from bone marrow to the periphery and induces systematic upregulation of TIMP-1
doi: 10.3324/haematol.2017.168799
Figure Lengend Snippet: Overview of the distribution pattern of MMP-9/TIMP-1 and MMP-2/TIMP-2 balances and MMP-9-positive cells upon challenge with lipopolysaccharide. Induction of endotoxemia results in the migration of MMP-9-positive polymorphonuclear cells out of the bone marrow to accumulate primarily in lungs and liver. This release of MMP-9 is meanwhile counteracted by a systematic upregulation of TIMP-1. In contrast, MMP-2 protein levels remain similar while TIMP-2 is generally reduced. NE; neutrophil elastase.
Article Snippet: TIMP-1 and
Techniques: Migration

Journal: Cell and Tissue Research
Article Title: MMP2 and acrosin are major proteinases associated with the inner acrosomal membrane and may cooperate in sperm penetration of the zona pellucida during fertilization
doi: 10.1007/s00441-012-1429-1
Figure Lengend Snippet: a Gelatin zymograms of detergent extracts ( RIPA ) of SSpH reveal MMP and serine proteinases as components of the IAM. Blockage of the 72-kDa enzymatic activity with GM6001 (Ilomastat) ( lane 2 ) indicates a metalloprotease and blockage of the 35-kDa enzymatic activities by trypsin inhibitor ( lane 3 ) indicates serine proteases. Lane 4 , a positive control, is loaded with a trophoblast cell medium containing both MMP2 (72 kDa) and MMP9 (92 kDa). b Gelatin zymogram without (control) and with (block) a cyclic disulfide bonded peptide (CTTHWGFTLC) on RIPA extracts of SSpH ( lane 1 ) and trophoblast media ( lane 2 ), containing both MMP2 and MMP9 enzymatic activities. c Immunoblotting verification that acrosin is responsible for serine protease activity in detergent extracts of SSpH. Anti-acrosin antibody detects proacrosin in western blots of whole bull epididymal sperm ( lane 2 ) and is blocked when preincubated with the peptide it was raised against ( lane 1 ). Sonication of whole sperm causes proacrosin cleavage into its active forms as indicated both in the resultant sonication supernatant ( lane 3 ) and in SSpH ( lane 4 ). The 2 % NP-40 (non-ionic detergent) extract of SSpH ( lane 5 ) is less efficient in stripping SSpH of proacrosin/acrosin than 2 % SDS ( lane 4 ). Lane 6 is a gelatin zymogram of the NP-40 extract loaded in lane 5 , confirming that the enzymatic activities found at the 35-kDa level are due to acrosin. d Immunoblotting verification that both MMP2 and Proacrosin/Acrosin are constituents of the sonicated bull sperm head. Freeze–thawed sperm ( lane 1 ) were sonicated and separated by centrifugation into three fractions: supernatant ( lane 2 ), SSpH ( lane 3 ), and tails ( lane 4 ). The sperm heads were then extracted with SDS ( lane 5 ) and compared with pellet ( lane 6 ). The upper part of the western blot was probed with anti-pMMP2, while the bottom part below the demarcating line was probed with polyclonal anti-bull acrosin antibody. e Immunoblots probed with anti-tMMP2 showing that MMP2 is present in bull, mouse and human spermatozoa (WS). Human recombinant MMP2, minus the pre-domain, is used as a positive control (rec. MMP2)
Article Snippet: Oocytes were pre-incubated in HTF with or without anti-pMMP2 (1:50), anti-tMMP2 (1:25), pre-immune anti-tMMP2 IgG purified antibody (1:10) or 40nM tissue inhibitor of
Techniques: Activity Assay, Positive Control, Control, Blocking Assay, Western Blot, Sonication, Stripping Membranes, Centrifugation, Recombinant

Journal: Cell and Tissue Research
Article Title: MMP2 and acrosin are major proteinases associated with the inner acrosomal membrane and may cooperate in sperm penetration of the zona pellucida during fertilization
doi: 10.1007/s00441-012-1429-1
Figure Lengend Snippet: Immunogold localization of MMP2 in bull spermatozoa and spermatid at end of cap phase. a In ejaculated sperm immunogold labeling, utilizing anti-tMMP2 antibody, is found through the apical ( AS ) and principal segments ( PS ) of the acrosome, a large portion associated with the IAM. See Suppl. Fig. b for preimmune control. Bar 0.2 μm. b In step 7-8 spermatid labeling with anti-pMMP antibody is seen along the inner acrosomal membrane ( arrows ) of the acrosome. AG acrosomic granule. Bar 0.2 μm
Article Snippet: Oocytes were pre-incubated in HTF with or without anti-pMMP2 (1:50), anti-tMMP2 (1:25), pre-immune anti-tMMP2 IgG purified antibody (1:10) or 40nM tissue inhibitor of
Techniques: Labeling, Control, Membrane

Journal: Cell and Tissue Research
Article Title: MMP2 and acrosin are major proteinases associated with the inner acrosomal membrane and may cooperate in sperm penetration of the zona pellucida during fertilization
doi: 10.1007/s00441-012-1429-1
Figure Lengend Snippet: Immunoperoxidase staining of MMP2 (with anti-tMMP2 antibody) in a testicular section of round spermatids in step 2–3 of bovine spermiogenesis. a As with acrosin, MMP2 immunostaining is found in the acrosomic granule ( arrows ) of proacrosomic and acrosomic vesicles before shifting to the acrosomal membrane during the cap phase of spermiogenesis. b Preimmune control. Bars 5 μm
Article Snippet: Oocytes were pre-incubated in HTF with or without anti-pMMP2 (1:50), anti-tMMP2 (1:25), pre-immune anti-tMMP2 IgG purified antibody (1:10) or 40nM tissue inhibitor of
Techniques: Immunoperoxidase Staining, Immunostaining, Membrane, Control

Journal: Cell and Tissue Research
Article Title: MMP2 and acrosin are major proteinases associated with the inner acrosomal membrane and may cooperate in sperm penetration of the zona pellucida during fertilization
doi: 10.1007/s00441-012-1429-1
Figure Lengend Snippet: Immunmoperoxidase staining of MMP2 in stages III and VII of the cycle of mouse seminiferous epithelium. a MMP2 immunostaining, utilizing anti-pMMP2 antibody, is associated with the acrosomic vesicles ( arrows ) of step 3 spermatids in stage III and the acrosomic cap of step 7 spermatids ( arrows ) in stage VII. The association of MMP2 with the acrosomic granule ( arrows ) of step 3 spermatids and the acrosomal membrane ( arrows ) of step 7 spermatids is seen to have a better advantage at higher magnification in the insets. Bars 10 μm. b Normal rabbit serum control
Article Snippet: Oocytes were pre-incubated in HTF with or without anti-pMMP2 (1:50), anti-tMMP2 (1:25), pre-immune anti-tMMP2 IgG purified antibody (1:10) or 40nM tissue inhibitor of
Techniques: Staining, Immunostaining, Membrane, Control

Journal: Cell and Tissue Research
Article Title: MMP2 and acrosin are major proteinases associated with the inner acrosomal membrane and may cooperate in sperm penetration of the zona pellucida during fertilization
doi: 10.1007/s00441-012-1429-1
Figure Lengend Snippet: Effect of anti-MMP2 antibodies and TIMP2 in mouse IVF. IVF control values were adjusted to 100 % for comparative purposes and experimental values obtained were adjusted accordingly. The block graphs depict the mean percentage of oocytes that were fertilized after incubation with sperm in IVF medium containing anti-pMMP2, anti-SPACA1, anti-tMMP2, or TIMP2, as compared to controls. Superscripts a and b denote a significant difference at P < 0.05 and error bars indicate standard error. In ( a ), only the pre-incubation medium with spermatozoa contained antibodies of which only part was transferred to the final IVF medium. In ( b ) both spermatozoa and oocytes were preincubated with antibodies and inhibitors that were combined in the final IVF medium. Thus, it is difficult to distinguish if the difference in the effectiveness of inhibition in ( a ) and ( b ) is due to anti-MMP2 antibody effectiveness or concentration of antibody in IVF media
Article Snippet: Oocytes were pre-incubated in HTF with or without anti-pMMP2 (1:50), anti-tMMP2 (1:25), pre-immune anti-tMMP2 IgG purified antibody (1:10) or 40nM tissue inhibitor of
Techniques: Control, Blocking Assay, Incubation, Inhibition, Concentration Assay

Journal: Experimental and Therapeutic Medicine
Article Title: Effect of elastin-derived peptides on the production of tissue inhibitor of metalloproteinase-1, -2, and -3 and the ratios in various endothelial cell lines
doi: 10.3892/etm.2015.2429
Figure Lengend Snippet: Concentration of (A) TIMP-1, (B) TIMP-2 and (C) TIMP-3 in the various endothelial cell lines (controls). Values are presented as the mean ± standard deviation. **P<0.01 and ***P<0.001, compared between groups. TIMP, tissue inhibitor of metalloproteinase.
Article Snippet: TIMP-1,
Techniques: Concentration Assay, Standard Deviation

Journal: Experimental and Therapeutic Medicine
Article Title: Effect of elastin-derived peptides on the production of tissue inhibitor of metalloproteinase-1, -2, and -3 and the ratios in various endothelial cell lines
doi: 10.3892/etm.2015.2429
Figure Lengend Snippet: Effects of various concentrations of κ-elastin on the TIMP-2 concentration in human endothelial cells derived from the (A) aorta, (B) iliac artery and (C) coronary artery. E1, E2, E3, E4 and E5, κ-elastin concentrations of 0.1, 0.4, 1.0, 2.5 and 5.0 µg/ml, respectively. Values are presented as the mean ± standard deviation. *P<0.05, **P<0.01 and ***P<0.001, compared between groups. N.S., not significant; TIMP, tissue inhibitor of metalloproteinase.
Article Snippet: TIMP-1,
Techniques: Concentration Assay, Derivative Assay, Standard Deviation